Highlighted Research / Work featured
Researchers develop smart fabric to neutralize nerve gas
By Fiona Haran 18 September 2017
The increasingly tough conditions that military personnel and emergency responders are faced with have led to the continuous evolution of smart textiles in military and emergency gear. Government support has created a demand that has helped the growth of the global smart textiles market in these sectors. And, thanks to the work of research institutes, advancements in material science present some exciting possibilities for future protective equipment.
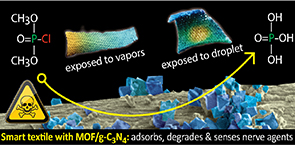
The City College of New York, for instance, has developed smart textiles with the ability to rapidly detect and neutralise nerve gas. The fabric consists of a cotton support modified with Cu-BTX metal organic framework (MOF)/oxidised graphitic carbon nitride detectors. Chemical engineer and Fulbright scholar, Teresa J Bandosz, who headed up the research, speaks to WTiN about what prompted the experiment. “The ultimate satisfaction for material scientists is that you make something that can be useful,” she says. “So, with this research, the aim was to deposit graphitic carbon nitride composites onto simple fabric to see if they could work, and to contribute somehow to the field of science and defence and personal protection.”
According to Bandosz, the graphitic carbon nitride composites were developed in the lab previously, and tested as nerve agent detoxification media and colorimetric detectors. Combining Cu-BTC and g-C3N4-ox resulted in a nanocomposite (MOFgCNox) of heterogeneous porosity and chemistry. Upon the deposition of MOFgCNox onto cotton textiles, a stable fabric with supreme photocatalytic detoxification ability towards the nerve gas surrogate, known as dimethyl chlorophosphate, was obtained. The detoxification process was accompanied by a visible and gradual colour change, which Bandosz says can be used for the selective detection of chemical warfare agents and for monitoring their penetration inside a protective layer. “These smart textiles adsorbed almost 7g of chemical warfare agent (CWA) surrogate per gram of Cu,” says Bandosz. “The superior performance was linked to the high dispersion of the MOF crystals on the fibres, and a specific texture promoting the availability of the active copper centres.”
When it came to selecting fabrics for testing, researchers were looking for fabrics that were “multi-functional” in order to achieve a “good catalytic phase”. Cotton was chosen for its large surface area. “It has oxygen groups on the surface for the position of our active phase,” says Bandosz. “Plus, the white colouring of cotton helps with sensing.” Potential applications of the smart textiles range from chemical suits and emergency response and military uniforms, to fabrics covering the actual military equipment, according to Bandosz, who has previously carried out research at the University of Malaga in Spain to help alleviate global warming and solve energy problems.
Further research
Bandosz is now seeking funding for additional research which, she reveals, will experiment with activated carbon fibres. “We found that if we’re able to modify them to deposit our active phase, we could bring more synergistic effects to the performance, as activated carbon textiles are also porous. They present an interface between our catalytic phase to aid performance, while the porosity inside the fibres can help by storing the composition products.” She continues: “We have broad experience with carbon, so we know how to make it active. More modification of the carbon fibres could bring more activity to the end product which could contribute to the oxidisation of the agents.” The team also aims to explore other compositions of the active phase. “We would like to find more synergy between the fabric support and the active phase; this is why we’re looking at porous fabrics,” says Bandosz.
The study appears in the journal Nanoscale Horizons.
A groundbreaking development has the potential to thwart chemical warfare agents: smart textiles
https://www.sciencedaily.com/releases/2017/08/170816134733.htm
From the lab of City College of New York chemical engineer and Fulbright Scholar Teresa J. Bandosz comes a groundbreaking development with the potential to thwart chemical warfare agents: smart textiles with the ability to rapidly detect and neutralize nerve gas.
The fabric consists of a cotton support modified with Cu-BTC MOF/oxidized graphitic carbon nitride composites. The latter were developed in the lab previously and tested as nerve agent detoxification media and colorimetric detectors.
Combining Cu-BTC and g-C3N4-ox resulted in a nanocomposite (MOFgCNox) of heterogeneous porosity and chemistry. Upon the deposition of MOFgCNox onto cotton textiles, a stable fabric with supreme photocatalytic detoxification ability towards the nerve gas surrogate, dimethyl chlorophosphate, was obtained.
The detoxification process was accompanied by a visible and gradual color change, which Bandosz said can be used for the selective detection of chemical warfare agents and for monitoring their penetration inside a protective layer.
“These smart textiles adsorbed almost 7g of CWA surrogate/its detoxification products per gram of Cu. The superior performance was linked to the high dispersion of the MOF crystals on the fibers, and a specific texture promoting the availability of the active copper centers,” said Bandosz, who is seeking funding for additional research.
Journal Reference:
- Dimitrios A. Giannakoudakis, Yuping Hu, Marc Florent, Teresa J. Bandosz. Smart textiles of MOF/g-C3N4 nanospheres for the rapid detection/detoxification of chemical warfare agents. Nanoscale Horiz., 2017; DOI: 10.1039/C7NH00081B
Story Source:
Materials provided by City College of New York. Note: Content may be edited for style and length.
Assignation as Editor at Journal of Colloid and Interface Science, Elsevier
PUBLISHER’S NOTE
We are pleased to welcome Professor Teresa J. Bandosz as Editor for the Journal of Colloid and Interface Science. Teresa J. Bandosz will reinforce the existing team of Editors consisting of Darsh Wasan (Editor in Chief), Julian Eastoe, Arthur Hubbard, Arturo Lopez-Quintela, Max Lu, Martin Malmsten, and Donguan Zhao. Due to the success of the journal and the strong increase in submissions the appointment of an additional Editor was needed.
Professor Baondosz leads a research group at the Department of Chemistry, The City College of New York, New York, USA. Her current research interests include: Environmental Application of Adsorption, Modification of materials, New sorbents and catalysts, Surface characterization, Methane/natural gas/hydrogen storage, Adsorption/desorption phenomena, Gas separation, Deep desulfurization of fuels, Catalytic photooxidation, Graphite oxide based composite adsorbents, Graphene/MOF composites as adsorbents, Gas sensors, Carbon materials based supercapacitors for energy storage.
She has been the author or co-author of 300 publications in peer reviewed journals which have been cited well over 7,000 times and she has an h-index of 41.
We are confident that, with her research focus and her experience in the field, Teresa will contribute to the existing reputation of our journal and strengthen its position in the scientific community. We wish her every success and personal satisfaction in her new position.
Rob van Daalen, Publisher Journal of Colloid and Interface Science
Amsterdam, The Netherlands
Journal of Colloid and Interface Science 414 (2014) A1
read the publisher’s note: http://dx.doi.org/10.1016/j.jcis.2013.11.001
Marie Roberts visits the
Prof. Teresa J. Bandosz lab
Marie Roberts visits Dr. Teresa Bandosz in her lab at City College as part of the Ligo Project Art of Science program.
Dr. Bandosz explains her interest in air purification and decontamination, as well as the work that is done in her lab and its real life applications.
Roberts experiments with the same carbon that Dr. Bandosz uses in her lab to create drawings of people from the lab.
Artist Marie Roberts with members of the T. J. Bandosz lab
Secret Weapon Against the Smell of Sludge? Sludge
http://www.nytimes.com/2005/02/21/science/21sludge.html?pagewanted=1&_r=0
Published: February 21, 2005

Ruby Washington/The New York Times – Teresa J. Bandosz is New York’s unofficial odor warden, monitoring how treatment plants keep odors from getting out and figuring out new ways to reduce the smell.
By ANTHONY DePALMA
Every city is saddled with aspects of urban life that are as undesirable as they are unavoidable – things, for instance, like sludge, and the stink that goes with it. Most people do not want to even think about how a city the size of New York gets rid of 1.2 billion gallons of everything that goes down the toilet, drain or gutter every day, and they certainly do not want to smell it.
And on most days, at most times, they do not, because sewage treatment plants are buttoned up tighter than a walk-in cigar humidor and people like Teresa J. Bandosz are there to make sure that what goes on inside stays inside.
Dr. Bandosz, a research scientist and professor of chemistry at City College, is New York’s unofficial odor warden. With a small team of student researchers and a technician, she monitors how well carbon filters at the treatment plants keep foul odors from spewing into the air.
Dr. Bandosz has also been trying to figure out how to make the smell-scrubbing process more efficient. Like a modern-day Edison, she has experimented with an odd assortment of materials to make the best filter. She has tried scrap paper and recycled plastic, then moved on to coal before going out on a limb and trying the pits from olives and peaches after she and her assistants ate around them.
Now she thinks she has come up with just about the perfect material for taking the stink out of sludge – sludge itself. She starts with fertilizer pellets that the New York Organic Fertilizer Company in the Bronx makes from the city’s treated sludge.
In an oxygen-free container, she heats the tiny pellets to more than 1,700 degrees, which helps them filter out offending compounds, primarily hydrogen sulfide, more efficiently. Each piece of treated sludge becomes like a microscopic sponge, filled with cavities that absorb the stuff that gives sludge its awful smell. “We ran the tests and we’ve had fantastic results,” said Dr. Bandosz, 44, who came to the United States from Poland in 1991. She said she has wanted to be a chemist since a teacher showed her the invisible world of compounds and molecules when she was 12.
“You had to use your imagination to link what you knew with what you couldn’t see,” she said.
continued………. read more: http://tbandosz.com/?page_id=530
Sewage’s Toxic Smell, Smothered by Coffee
http://www.nytimes.com/2012/02/21/science/sewages-toxic-smell-smothered-by-coffee.html?_r=0
Published: February 21,2012
By SINDYA N. BHANOO
Coffee lovers around the world can rejoice: The piles of grounds they discard could help rid the world of the toxic smell of sewage. Writing in The Journal of Hazardous Materials, researchers at the City University of New York report thatcoffee grounds can absorb hydrogen sulfide gas, a big part of what makes sewage smell so terrible.
 Today, activated carbons or porous coals are used in treatment facilities to draw hydrogen sulfide from sewage. But when coffee grounds are transformed into activated carbon, the researchers found that they sop up sulfur particularly well. That’s because of a key ingredient in coffee: caffeine.
Today, activated carbons or porous coals are used in treatment facilities to draw hydrogen sulfide from sewage. But when coffee grounds are transformed into activated carbon, the researchers found that they sop up sulfur particularly well. That’s because of a key ingredient in coffee: caffeine.
Caffeine contains nitrogen, which increases carbon’s ability to eliminate sulfur from the air, said Teresa J. Bandosz, a chemist and chemical engineer at CUNY and an author of the report. To carbonize the coffee grounds, she and her colleagues mixed the grounds with water and zinc, and then dried the mixture in an oven. Dr. Bandosz hopes that entrepreneurs might take the research and turn it into a business.
A coffee drinker herself, Dr. Bandosz came up with the idea because she throws out piles of coffee grounds. “Fresh coffee would work even better — it has more caffeine,” she said. “But it is not economical.”
read more: http://www.nytimes.com/2012/02/21/science/sewages-toxic-smell-smothered-by-coffee.html?_r=0
Carbonized coffee grounds remove foul smells
http://www.rdmag.com/news/2012/02/carbonized-coffee-grounds-remove-foul-smells
Published: February 22, 2012
For coffee lovers, the first cup of the morning is one of life’s best aromas. But did you know that the leftover grounds could eliminate one of the worst smells around—sewer gas?
In research to develop a novel, eco-friendly filter to remove toxic gases from the air, scientists at The City College of New York (CCNY) found that a material made from used coffee grounds can sop up hydrogen sulfide gas, the chemical that makes raw sewage stinky.
Teresa Bandosz, PhD, CCNY professor of chemistry and chemical engineering, develops and tests materials that scrub toxic gases like hydrogen sulfide from air in industrial facilities and pollution control plants. Much like the grains of charcoal packed into the filter of a tabletop water pitcher, her filters use a form of charcoal called “activated carbon.”
Carbon producers already use materials like coal, wood, peat, fruit pits, and coconut shells to make filters. Bandosz realized that our modern coffee culture could supply an abundant source of eco-friendly organic waste. But coffee grounds also come equipped with a special ingredient that boosts their smell-fighting power.
Caffeine, the stimulant that gives coffee its energy jolt, contains nitrogen. This element cranks up carbon’s ability to clean sulfur from the air, a process called adsorption. “We should not neglect the natural biomass that is rich in this element,” she and colleagues assert in the Journal of Hazardous Materials. The National Science Foundation (NSF) and the Army Research Office funded the research.
Usually, making carbon adsorbents more reactive to toxins requires treating the original with a nitrogen-rich chemical such as ammonia, melamine, or urea, the main nitrogen-containing substance in mammal urine. “All of these,” the researchers note, “significantly increase the cost of adsorbents.”
To make their new filter, Bandosz and her colleagues carbonized old coffee grounds, essentially turning them into charcoal.
To do so, they prepared a slurry of coffee grounds, water and zinc chloride, a chemical “activator.” The team then dried and baked the mixture at temperatures of up to 800 C. The process of activation fills the carbon with scores of minute holes about 10 to 30 A in diameter, roughly equivalent to 10 to 30 hydrogen atom-widths across. These densely packed pores are blanketed with nitrogen, perfect to capture hydrogen sulfide molecules passing through.
Hydrogen sulfide gas isn’t just a smelly nuisance for sewage plant neighbors; it can be deadly. Human noses are so sensitive to the rotten-egg scent of this toxin that it can overwhelm the sense of smell, Bandosz explained. “When someone is exposed to high concentrations of hydrogen sulfide, the nose will stop detecting it,” she said. “There have been cases in which workers died of hydrogen sulfide exposure in sewer systems.” Bandosz suspects that the coffee-based carbon could also separate out other pollutants from the air and water.
With the ubiquitous motto to “Reduce, Reuse, Recycle,” and coffee-ground carbon’s special affinity for a toxic gas, Bandosz hopes coffee grounds can be commercially developed into the next green waste filter. For now, however, she recycles them on her own: “I put them outside under the plants in my garden, especially those that like acidic soil,” she said. They are a great fertilizer, of course, packed as they are with nitrogen-rich caffeine.
read more: http://www.rdmag.com/news/2012/02/carbonized-coffee-grounds-remove-foul-smells
Sludge to Suppress Sewage Smells
http://www.nydailynews.com/archives/news/sewers-flush-cash-article-1.658258#ixzz2uTFUyj1G
Published: February 14, 2005
BY CLEM RICHARDSON / NEW YORK DAILY NEWS
TERESA BANDOSZ looks at sewage and sees air freshener. And money, lots of money, that now is being flushed down the drain. The 44-year-old chemistry professor at the City College of New York is researching ways to improve the quality of air emitted from sewage-treatment plants. Bandosz has found a solution in the sewage itself. As necessary as sewer-treatment plants are to the continued health of a city and its population, few neighborhoods welcome their presence and often campaign to keep them out or close them down. New York City has 14 plants scattered across the boroughs, the most prominent being the North River Plant, on the Hudson between W. 137th and W. 145th Sts., which processes 200 million gallons of waste a day. Essentially, sewer plants remove liquid from waste, purifying the remaining solids into a nutrient-rich fertilizer called sludge. Lots of sludge. One plant can generate up to 10 million dry tons of the stuff a day, Bandosz said. “The problem is, we have to find something to do with it because it can no longer be dropped in the ocean,” Bandosz said. More on that later. One reason people dislike sewer plants as neighbors is that they often smell, despite millions of dollars spent on chimneys, air-filtration systems and other techniques to limit odors. The culprit is a chemical, hydrogen sulfide, which has a noxious smell that often survives all attempts to eliminate it. Bandosz, who holds a chemical engineering degree from the University of Mining and Metallurgy in her native Poland, has been working on the problem with the city Department of Environmental Protection since joining City College in 1996. DEP oversees the city sewers. Air in a plant like the North River is filtered through an internal purification system and released from high towers. Part of that system sends the air through a layer of carbon, which interacts with the hydrogen sulfide, breaking down much of the chemical into its component parts. Simple enough so far. One problem is that the current process used by the city requires 1,400 tons of activated carbon, enough to fill 14 railroad cars. The carbon now being used, a virgin variety produced by burning coconut shells, costs $1 a pound and the supply needs to be replenished constantly. Bandosz was one of the experts who persuaded the city to switch to virgin carbon years ago in a bid to cut down on fires caused when impurities in the lesser grade of carbon then in use would cause the hydrogen and sulfur produced in the process to ignite. Still, the virgin carbon is an added expense, “and they still have to get rid of the sludge,” Bandosz said. Bandosz’s idea is to burn the sludge at 1,720 degrees Fahrenheit to create an organic carbon that has proven even more effective than the virgin carbon now used in breaking down hydrogen sulfide. This organic carbon breaks more of the hydrogen sulfide into its component parts, producing sulfur that could be disposed of more easily. “We have to get rid of the sludge anyway,” Bandosz said. “We can use sludge to treat sludge and save a tremendous amount of money at the same time.
” Bandosz’s system, which uses alternate layers of virgin carbon and sludge carbon, is already being tested in one of the city’s sewer plants. She is in the process of applying for a patent for the process. crichardson@nydailynewscom SIDEBAR Teresa Bandosz was headed to Argentina after graduating from college but came to the U.
Read more: http://www.nydailynews.com/archives/news/sewers-flush-cash-article-1.658258#ixzz2uTFUyj1G